Colombia Is Leading the IVF Momentum in Latin America — And Mondo IVF Is Setting the Standard in Reproductive Logistics

Executive Summary
Colombia has become one of the most strategic IVF destinations in Latin America, driving a significant increase in international reproductive treatment cycles and cross-border biological material transport.
This week, three international Mondo IVF couriers converged in Colombia from different global cities to execute coordinated hand-carry transport of reproductive samples under strict chain of custody and full international regulatory compliance.
This operation reflects a broader market reality:
- IVF treatment demand in Colombia is accelerating
- International intended parents require secure embryo and gamete transport
- Clinics demand logistics partners operating at clinical-grade precision
- Regulatory oversight across borders is becoming more complex
In reproductive logistics, shipment is not the objective.
Risk mitigation, regulatory integrity, and custody control are.
Colombia: A Strategic IVF and Assisted Reproduction Hub in Latin America
Why Is Colombia Experiencing Accelerated IVF Growth?
Colombia has positioned itself as a leading destination for:
- In vitro fertilization (IVF)
- Oocyte donation programs
- Embryo transfer cycles
- Fertility preservation
- Cross-border reproductive care
Cities like Bogotá, Medellín, Cali, Cartagena, and Bucaramanga are now firmly on the global fertility map — and with that growth comes the need for logistics partners capable of operating at the same level of precision and responsibility as the clinics themselves.
The growth is driven by:
- Internationally recognized fertility clinics
- Advanced embryology laboratories
- Competitive treatment accessibility
- Strong medical tourism infrastructure
- Increasing regulatory alignment with international standards
With growth comes a parallel demand: high-precision reproductive logistics.
Transporting cryopreserved embryos, oocytes, or sperm internationally is not comparable to general medical courier services. The risk profile is materially higher, and the tolerance for error is effectively zero.
What Is Reproductive Logistics — and Why Is It Different?
Reproductive logistics refers to the specialized transport of cryopreserved biological material used in assisted reproduction, including:
- Frozen embryos
- Cryopreserved oocytes
- Semen samples
- Reproductive tissue
Unlike general medical shipping, IVF transport requires:
- Continuous temperature stability (cryogenic conditions at approximately -196°C in vapor-phase liquid nitrogen tanks)
- Documented chain of custody at every transfer point
- Compliance with international health authorities and aviation regulations
- Direct coordination with fertility clinics and laboratory directors
- Real-time monitoring and communication
This is not freight forwarding.
It is clinical risk management in transit.
The Mondo Standard in Action: Coordinated International IVF Transport
Operational Snapshot
This week in Colombia, three international routes converged under a unified protocol.
Operation Included:
- Dedicated hand-carry medical courier (no cargo shipment)
- Vapor-phase LN2 dry shipper transport (~ -150°C to -196°C stability range)
- Continuous chain of custody documentation
- Full compliance with IATA Dangerous Goods Regulations (when applicable)
- Alignment with importing/exporting country health authority requirements
- Direct coordination with fertility clinics and local authorities
- Real-time communication throughout transit
Each route was executed under protocol-driven precision.
Because in reproductive sample transport, temperature deviation, documentation error, or custody break are not operational inconveniences — they are irreversible events.
Technical Summary: Risk Control in International IVF Transport
Before entering regulatory detail, it is important to clarify the primary risk variables in cross-border reproductive transport:
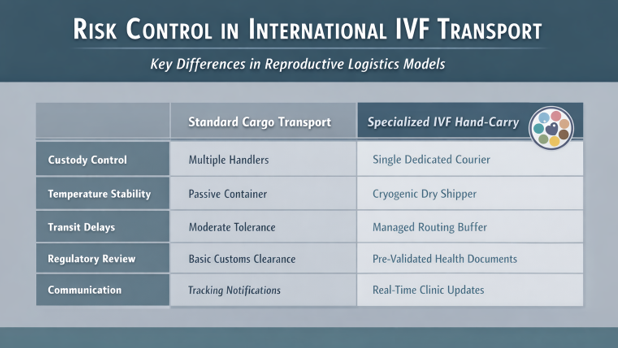
Full International Regulatory Compliance
International IVF transport requires compliance across multiple layers:
- Export country health authority requirements
- Import country reproductive material authorization
- Airline-specific dangerous goods policies
- Airport security regulations
- Customs documentation accuracy
- Chain of custody signatures at each checkpoint
In Latin America, regulatory interpretation can vary by jurisdiction. Colombia’s increasing IVF volume demands logistics partners who understand:
- Documentation sequencing
- Advance permit requirements
- Local clinic coordination
- Airport-level procedural realities
Compliance is not reactive.
It is pre-engineered before departure.
More Than Growth — A Shared Ethical Standard
What made this coordinated moment in Colombia meaningful was not simply operational expansion.
It was alignment.
Three couriers from different global cities.
Three distinct routes.
One unified protocol.
Meeting in an airport and recognizing the same standard:
- The same respect for the cryogenic tank in hand
- The same meticulous attention to documentation
- The same understanding that every shipment represents a family
- The same refusal to compromise on chain of custody
Reproductive logistics is human before it is logistical.
And human systems require shared ethics.
Decision-Making Criteria for Clinics and Intended Parents
For fertility clinics and international patients evaluating a reproductive logistics partner, the criteria are not cosmetic.
They are structural:
- Is transport cargo-based or dedicated hand-carry?
- Is chain of custody continuous and documented?
- Is the dry shipper validated before departure?
- Is there real-time communication during transit?
- Is regulatory compliance proactively engineered?
- Is there direct coordination with both origin and destination clinics?
The cost of choosing incorrectly is not financial alone.
It is biological and irreversible.
Colombia’s IVF Growth Requires Elevated Transport Standards
Colombia is expanding its role in international assisted reproduction.
Demand is increasing.
Cross-border cycles are rising.
Global intended parents are choosing Colombian clinics.
Standards must rise proportionally.
The benchmark in biological material transport is not automatic.
It is built daily through:
- Protocol
- Precision
- Accountability
- Human responsibility
And it is being executed, route by route, by Mondo IVF.
FAQ — IVF Embryo Transport
What temperature must embryos be transported at?
Cryopreserved embryos are transported in vapor-phase liquid nitrogen dry shippers, typically maintaining internal temperatures between approximately -150°C and -196°C to preserve cellular integrity.
Is cargo shipment acceptable for IVF samples?
While technically possible under certain conditions, cargo shipment introduces multiple custody handoffs and higher delay risk. Dedicated hand-carry medical courier models significantly reduce exposure variables.
What regulatory bodies are involved in international embryo transport?
Depending on the route, transport may involve:
- National health authorities (export/import permits)
- Customs agencies
- Civil aviation authorities
- Airline dangerous goods departments


0 comentarios