IVF Embryo, Egg, and Sperm Transport: Safety Standards, -196°C Temperature Control, and Hand-Carry vs Cargo Explained

Executive Summary
Transporting embryos, oocytes (eggs), and sperm requires IVF cryogenic transport protocols, strict regulatory compliance, and continuous temperature monitoring at -196°C.
Choosing between standard cargo shipping and a hand-carry IVF courier service directly impacts biological integrity, legal compliance, and chain of custody security.
This guide explains:
- How embryo transport works
- Why -196°C temperature control is critical
- Why X-ray exposure must be avoided
- What defines a premium IVF transport service
- Why hand-carry is considered the gold standard
What You Need to Know Before Hiring an IVF Embryo Transport Service
The journey to building a family through assisted reproduction is deeply emotional. When transporting embryos, eggs, or sperm across state lines or international borders, safety, compliance, and temperature stability are non-negotiable.
Before hiring an IVF embryo transport service, it is essential to evaluate three pillars:
- Cryogenic technology standards
- Regulatory compliance (FDA, TSA, IATA)
- Transport modality (hand-carry vs cargo)
Not all IVF transport providers operate at the same level of safety or regulatory precision.

A premium IVF cryogenic transport service combines speed, monitoring, compliance, and uninterrupted custody.
When evaluating a provider, look for:
Advanced Cryogenic Technology
- Medical-grade MVE dewars
- Vapor-phase liquid nitrogen
- Stable -196°C embryo transport temperature
- No free-flowing liquid nitrogen
Speed and Efficiency
- Same-day or 24-hour mission completion
- Minimized transit exposure time
- Active coordination between fertility clinics
Continuous Temperature Monitoring
- Digital data loggers
- Reporting intervals every 20 minutes
- Temperature validation documentation
- Cryogenic stability verification
Dedicated 24/7 Support
- Real-time communication
- Clinic-to-clinic coordination
- Customs pre-clearance management
A true IVF logistics company prioritizes biological integrity over shipping convenience.
Global Security Protocols and Compliance in IVF Transport
The international transport of human reproductive tissue is highly regulated to protect biological viability and prevent disease transmission.
Professional IVF transport services must comply with:
- Food and Drug Administration – 21 CFR Part 1271
- Transportation Security Administration
- International Air Transport Association
Critical safety protocols include:
- Strict prohibition of X-ray exposure
- Manual security inspection (“hand search”)
- Upright cryogenic tank orientation
- Continuous chain of custody documentation
- Hand-carry only (never checked as luggage)
These measures ensure regulatory compliance and embryo viability during international IVF transport.
What Is IVF Cryogenic Transport?
IVF cryogenic transport is the process of moving frozen embryos, oocytes, and sperm at a constant -196°C using vapor-phase liquid nitrogen inside a medical-grade dry shipper.
This process:
- Halts cellular metabolism
- Preserves DNA structure
- Prevents ice crystal formation
- Maintains long-term viability
What Temperature Are Embryos Transported At?
Embryos are transported at -196°C (-320.8°F) — the boiling point of liquid nitrogen.
Maintaining this temperature ensures:
- Genetic stability
- Structural cell preservation
- Long-distance transport safety
Even brief temperature deviations may compromise viability.
Why Must Embryos Avoid X-Ray Exposure?
X-ray radiation is ionizing and can:
- Damage DNA strands
- Cause chromosomal disruption
- Reduce implantation success rates
For this reason, professional IVF hand-carry couriers request manual TSA inspection to bypass X-ray scanners entirely.
What Is a Dry Shipper and How Does It Work?
A cryogenic dry shipper is a vacuum-insulated container that absorbs liquid nitrogen into a porous core.
It:
- Maintains vapor-phase -196°C
- Prevents nitrogen spills
- Meets aviation safety standards
- Protects embryos during air transport
The tank must remain upright to preserve vacuum integrity and nitrogen retention.
Regulatory Compliance in Embryo Transport
Transporting embryos internationally requires strict regulatory compliance and data privacy safeguards.
In addition to physical security, IVF transport services handle Protected Health Information (PHI) and must operate under strict privacy frameworks.
Professional providers must ensure:
- HIPAA compliance
- Business Associate Agreements (BAAs)
- Encrypted patient data systems
- 24–48 hour breach reporting protocols
- Secure documentation handling
Full regulatory compliance protects both biological material and patient confidentiality during international embryo transport
Hand-Carry vs Standard Cargo: What Is the Difference?
When transporting frozen embryos internationally, intended parents and fertility clinics typically face two options: standard cargo shipping or a specialized hand-carry IVF courier service.
Standard cargo treats reproductive material as freight, often involving multiple handlers, automated screening, and limited monitoring. In contrast, hand-carry services prioritize uninterrupted custody and direct supervision.
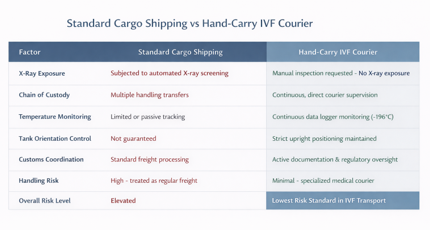
Standard cargo increases risk due to handling variability and automated X-ray screening.
Why Hand-Carry Is the Gold Standard in IVF Transport
The hand-carry IVF courier model represents the highest standard in reproductive logistics.
In this modality:
- A specialized onboard courier personally escorts the cryogenic tank.
- The tank never leaves physical custody or line of sight.
- Manual inspection replaces X-ray scanning.
- Upright positioning is constantly maintained.
- Real-time coordination reduces customs delays.
Unlike cargo shipping, hand-carry transport eliminates:
- Automated X-ray exposure
- Multiple custody transfers
- Mishandling risks
- Unmonitored environmental changes
For families investing emotionally and financially in IVF treatment, hand-carry embryo transport offers unmatched safety, compliance, and peace of mind.
Safely Delivering Your Future with Mondo IVF

Mondo IVF operates exclusively under the hand-carry IVF courier model, combining cryogenic precision, regulatory compliance, and global coordination.
Logistics & Compliance
- Full adherence to FDA, TSA, and IATA standards
- Clinic-to-clinic coordination
- Chain of custody documentation
- 21 CFR Part 1271 compliance
Global Presence
- Active operations across the U.S., Canada, EU, and Latin America
- Customs clearance expertise
- International embryo transport specialization
Absolute Privacy
- HIPAA-compliant data protection
- Mandatory Business Associate Agreements
- Secure patient information handling
Liability & Security
- Continuous -196°C vapor-phase transport
- MVE dewars
- Temperature data loggers reporting every 20 minutes
- Direct courier custody at all times
Your embryos, eggs, and sperm are never treated as cargo.
They remain under specialized professional supervision from origin to destination.
FAQ — IVF Embryo Transport
Is it safe to ship frozen embryos internationally?
Yes, when transported at -196°C using vapor-phase liquid nitrogen and under hand-carry supervision with regulatory compliance.
Can embryos go through airport X-ray machines?
No. X-ray radiation may damage DNA. Manual inspection must be requested.
How long can embryos remain in a dry shipper?
High-quality dry shippers can maintain -196°C for several days, depending on model and preparation.
What is the safest way to transport embryos?
Hand-carry IVF courier service is considered the safest modality due to continuous custody and elimination of X-ray exposure.

0 comentarios